A woman says weight loss injections turned her tongue jet black and left her needing her gallbladder removed.
Sarah-Jayne Crawford, 32, began using Wegovy in August 2024 after her weight reached 19 stone 9lbs (275lb).
The mum-of-one wanted to lose weight ahead of her wedding in November 2025. She ordered the injections through an online pharmacy, paying £199 ($269) for a month’s supply at a 0.25mg dose.
However, after 12 weeks — and a further £350 ($474) spent — she increased the dose to 1mg and says her health then deteriorated rapidly. Sarah reported “sharp pains” high in her abdomen and repeated vomiting, before waking in December to find her tongue had turned jet black.
She says the tongue discoloration came with intense mouth pain, difficulty eating spicy food, and unpleasant sulphur burps with an “egg” smell.
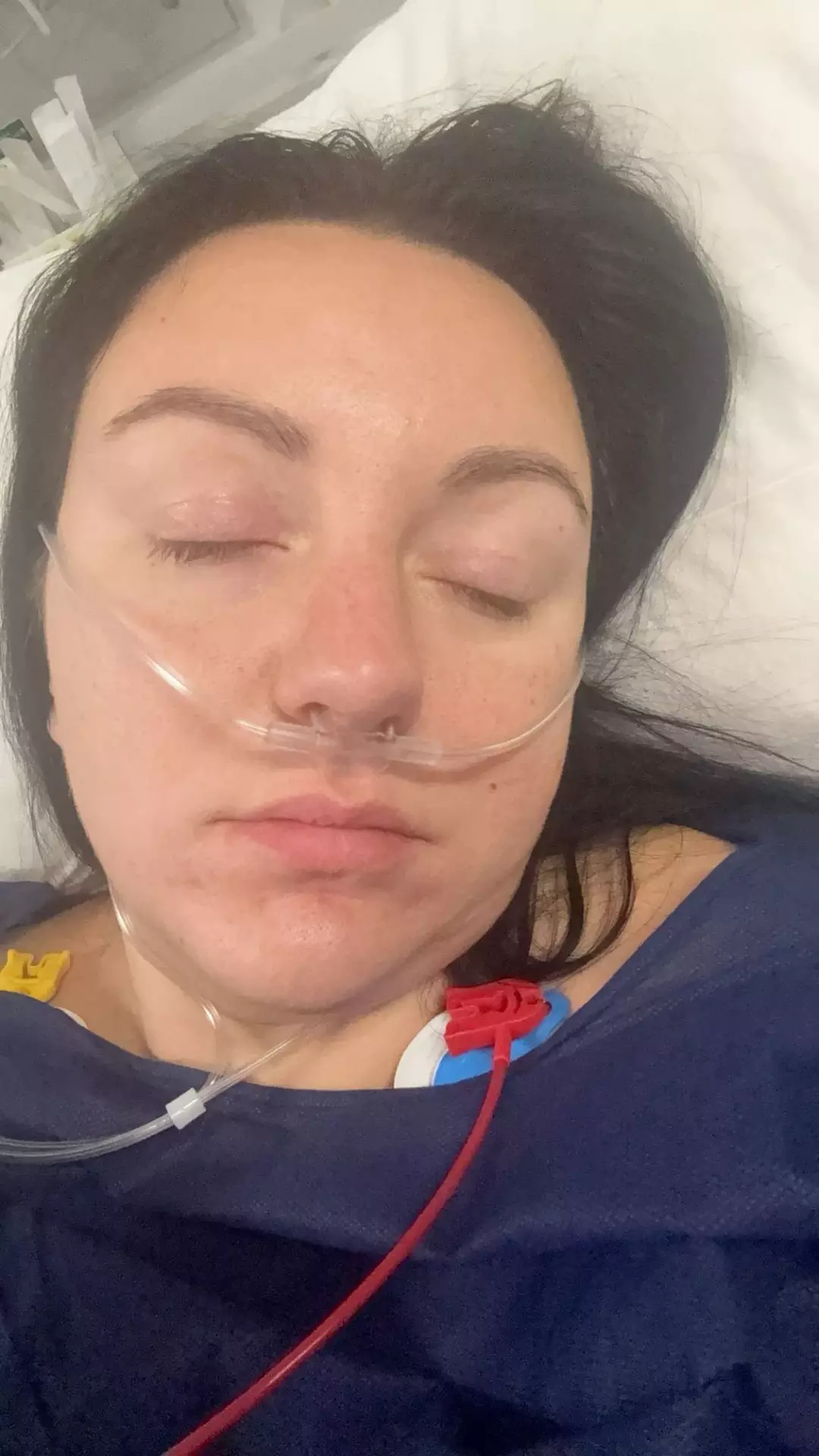
Worried by the sudden symptoms, she went to the emergency room. CT and MRI scans later identified “multiple” gallstones obstructing her bile duct, as well as diverticulitis (inflammation of small pouches in the wall of the large intestine). She then stopped taking the injections.
In February 2025, Sarah underwent a cholecystectomy to remove her gallbladder after the gallstones were confirmed.
Now off the medication and close to recovery, she says she remains confused by the black-tongue episode and is sharing her experience to encourage others to be aware of potential risks linked with weight-loss injections.
Sarah, a personal secretary for the NHS, from Antrim, Northern Ireland, said: “Wegovy was my last resort. I know a lot of people who are on the jabs and it worked like a dream for them.
“I had been battling my weight for what felt like forever and I was dying to slim before my wedding, but dieting, exercising and starving myself never worked. Before, I had heard stories about the dangers and my friend even joked about gallstones.
“But when the pain left me unable to leave my bed I knew something was up. The black tongue seemed like a cherry on top and a warning of what was to come.
“Never did I think this would happen to me.”
She says she had struggled with her weight for as long as she can remember, and that getting engaged in September 2022 pushed her to try to make a change.
Believing “traditional” approaches hadn’t worked for her in the past, Sarah says she paid £199 ($269) via an online pharmacy for a two-month supply of Wegovy at a 0.25mg dose.
“At the start, everything seemed fine,” she said. “My appetite slowly disappeared and I felt great for the wedding. But I wasn’t noticing a huge amount of change.”
But after 12 weeks, she says she began experiencing intense episodes of pain in the upper stomach area, along with vomiting. The discomfort then escalated into what she described as an “excruciating” trapped-wind sensation, leaving her confined to bed.
Within a day, she noticed the dark discoloration of her tongue.
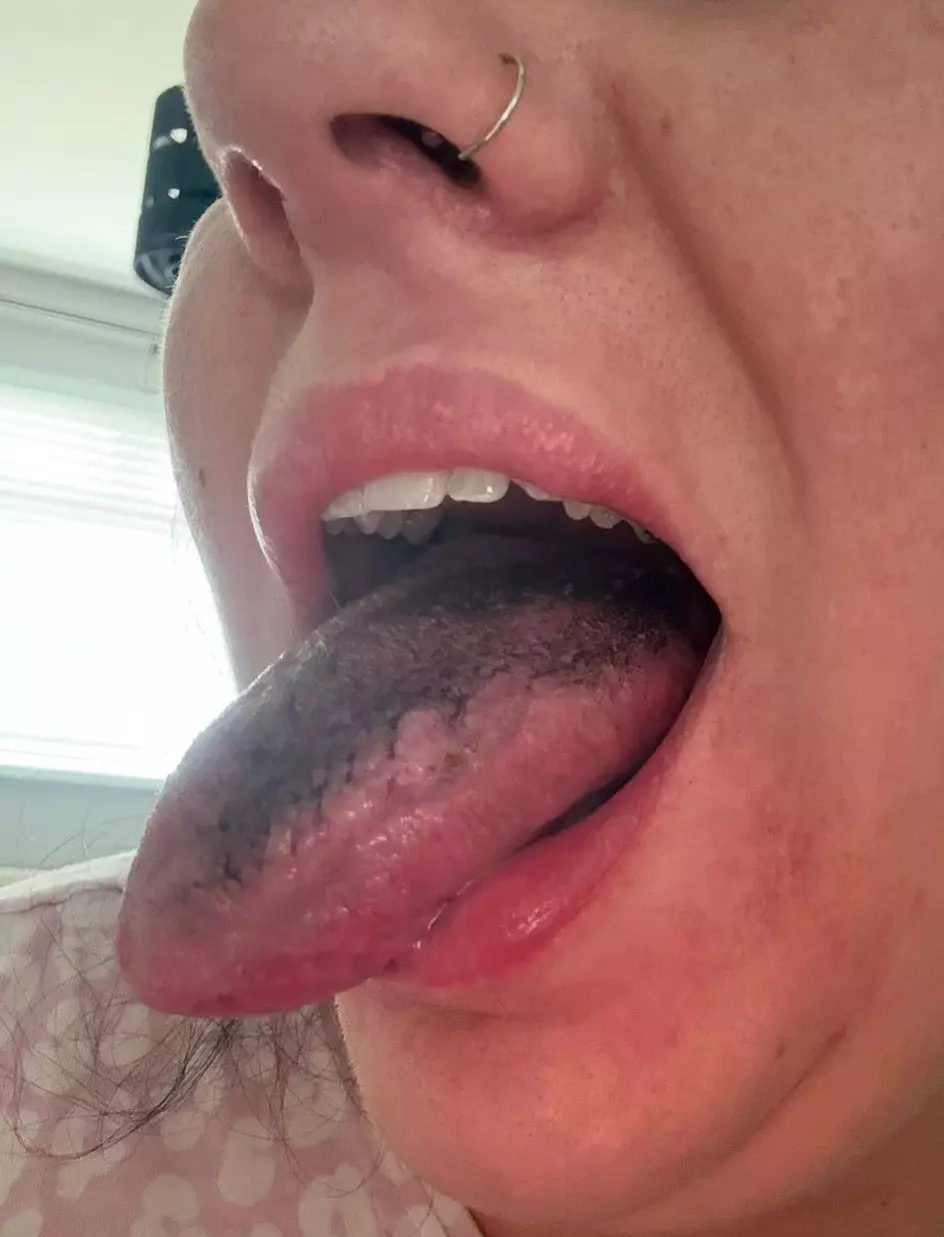
“It all happened so quickly,” Sarah said. “It went from really sharp pains that were enough to leave me bed bound.
“To having sulphur burps, that smelt of egg, and a black tongue. It was terrible.”
Sarah went on: “I have a child, so it was very difficult to take time off work. But the pain was so unbearable I couldn’t walk.
“I would rather give birth to five kids at the same time than go through with the pain again.”
According to the National Center for Biotechnology Information, semaglutide (the active ingredient in Wegovy, used to help with glucose control and weight loss) is associated with side effects including dry mouth, halitosis (bad breath), and sulphur-smelling burps due to slowed digestion — and it may also contribute to a black-coated tongue.
Sarah says she stopped the injections immediately and went to hospital, where emergency MRI and CT scans found multiple gallstones blocking her bile duct, placing her at risk of pancreatitis.
She was scheduled for a full cholecystectomy on February 10, 2025, and says her surgeon advised her to remain off Wegovy to proceed safely with anaesthetic.
“By this point, I was 18 stone and had hardly lost any weight,” she said. “The procedure was about an hour and I cracked on with the recovery.
“I’m slowly transitioning back to normal and eating more food now, but I’m still far from normal.”
She says that since discontinuing the injections in December 2024, her tongue has returned to its usual colour.

A spokesperson from Novo Nordisk, a Wegovy manufacturer, said issued a statment in the wake of Sarah’s experiences.
They said: “Patient safety is of the utmost importance to us at Novo Nordisk. To ensure healthcare professionals have a detailed understanding of our medicine’s safety profile, the adverse reactions we have observed in clinical trials are detailed in our medicine’s Summary of Product Characteristics (SmPC).
“In the registrational trials for this medicine, cholelithiasis (acute gallstone disease) was reported in 1.6% of patients and led to cholecystitis in 0.6% of people living with obesity treated with semaglutide 2.4 mg.
“For this reason, acute gallstone disease (cholelithiasis) is listed as a “common” potential adverse reaction for Wegovy (semaglutide injection) in the product’s UK SmPC and should be considered when patients are being evaluated for this medicine.
“As part of this work we also monitor reports of adverse drug reactions through routine pharmacovigilance.
“If you know of someone who might be experiencing side effects from any medicine, we would advise you to report it to your healthcare provider, and via the MHRA Yellow Card Scheme.”

